‘Lower is better’ underpins the management of low-density lipoprotein cholesterol (LDL-C) in the 2019 European Society of Cardiology/European Atherosclerosis Society (ESC/EAS) dyslipidaemia guidelines (1). Recent trials in very high-risk patients with ezetimibe and PCSK9 monoclonal antibody therapy provide robust support, showing incremental reduction in cardiovascular events at lower LDL-C levels (2 4), with no evidence to suggest a threshold for clinical benefit from LDL-C lowering (5). The available data also do not indicate safety issues associated with very low LDL-C levels attained with these efficacious therapies. Thus, the totality of evidence provides a strong basis for lowering LDL-C goals together with at least 50% reduction from baseline levels in high-risk and very-high-risk patients (1).
Why is this EAS Task Force statement needed?
Achieving these new LDL-C goals is a challenge for clinicians. The DA VINCI survey of lipid management in 18 European countries highlights this (6. Among secondary prevention patients, less than half were on high-intensity statin monotherapy, and of these only about one in five attained the 2019 ESC/EAS goals. While improving uptake of maximally tolerated high-intensity statin therapy is needed, most very-high-risk patients will not attain LDL-C goal with statin alone. Thus, as recommended by the guidelines, combination LDL-C-lowering therapy should become the rule for treating high-risk and very-high-risk patients with hypercholesterolaemia (1,7). Targeting highly efficacious LDL-C-lowering combination therapy to patients at highest risk who gain the most clinical benefit is also a cost-efficient strategy, subject to local reimbursement (8).
Clinicians need information on how best to implement these guideline recommendations in their routine practice. This new statement from an EAS Task Force provides practical guidance to improve LDL-C goal attainment aided by accessible algorithms (9).
Combination LDL-lowering paradigm
LDL-C management in high- and very-high risk patients should be guided by two important criteria: i) the starting LDL-C level, i.e., how far the patient is from the recommended LDL-C goal, and ii) the average LDL-C reduction that is achievable with each therapy. Thus, in patients with atherosclerotic cardiovascular disease (ASCVD) who are more than 50% from LDL-C goal, upfront high-intensity statin-ezetimibe combination therapy is recommended.
The Task Force recognises that there may be practical concerns about next approaches for add-on LDL-C lowering therapy, especially in a cost-limited environment. While PCSK9 inhibitors are undoubtedly highly efficacious in lowering LDL-C, their cost-effectiveness also needs to be taken into account. Pragmatically, individuals who are at highest risk with persistently elevated LDL-C levels should be preferably targeted. Thus, addition of a PCSK9 inhibitor may be considered in individuals who do not attain goal with the combination of ezetimibe and statin and have additional risk modifiers (Figure 1).
For patients with documented statin intolerance, in accordance with EAS recommended criteria (10), the combination of ezetimibe and bempedoic acid may be considered.
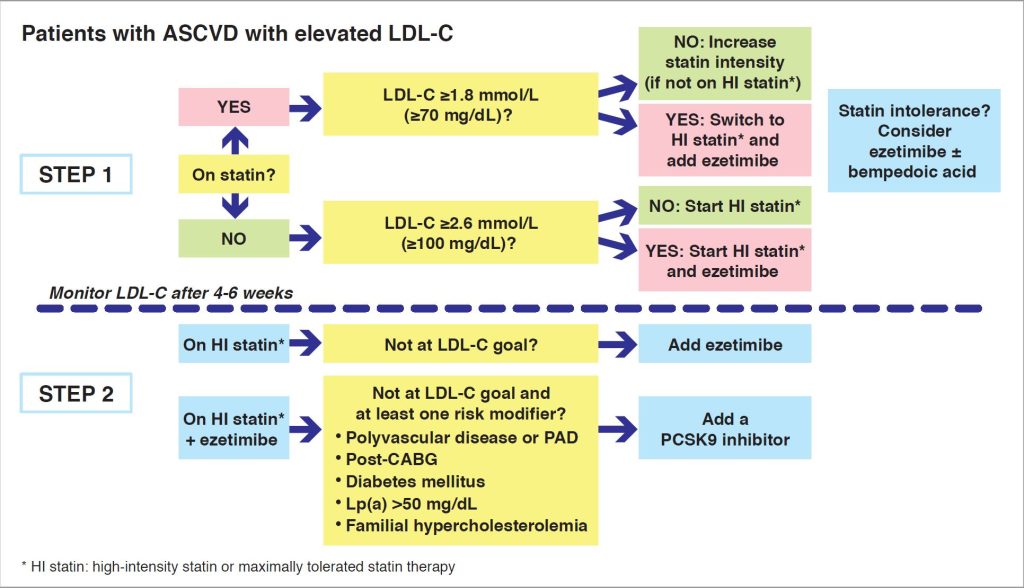
Figure 1. Algorithm for managing elevated LDL-C in secondary prevention
The 2019 ESC/EAS dyslipidaemia guidelines recognised the high risk associated with familial hypercholesterolaemia (FH), including those without ASCVD (1). In all FH patients, including those without ASCVD, combination statin-ezetimibe therapy should be the standard of care, with the addition of a PCSK9 (subject to reimbursement) for individuals with at least one risk modifier who are not at LDL-C goal (Figure 2) (1,11).
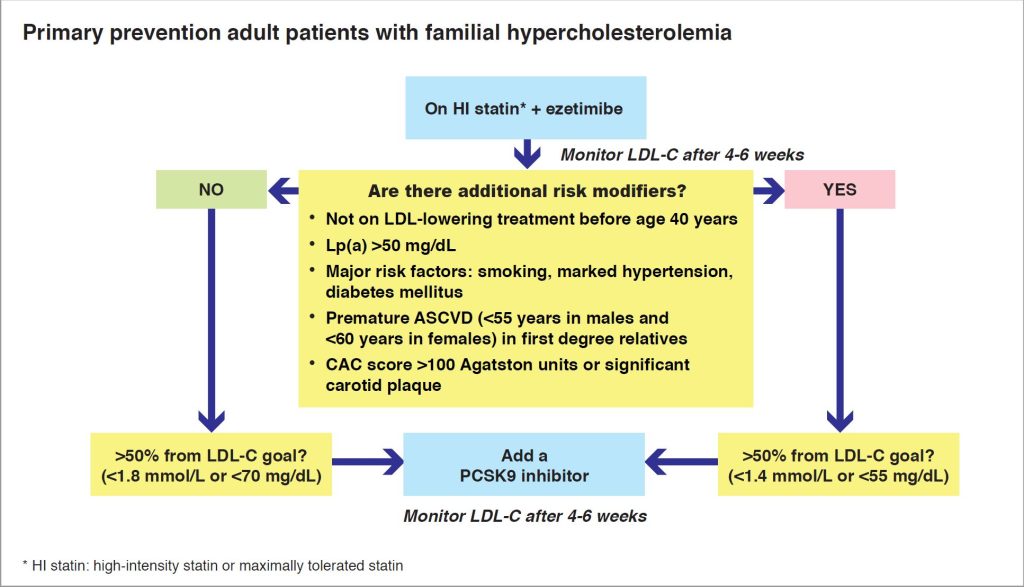
Figure 2. Algorithm for managing LDL-C in primary prevention FH patients
One question often posed by patients is whether LDL-C lowering nutraceuticals are an alternative to licensed pharmacotherapies (such as statins). Certainly, evidence supports the LDL-C lowering effects of whole foods such as nuts, pulses, oat cereal, psyllium, or foods supplemented with 2-3 g/day phytosterols (plant sterols or stanols), ideally in combination to provide LDL-C lowering efficacy comparable to that of low dose statin therapy (1,12). The evidence for individual LDL-C lowering nutraceuticals is more variable, best represented by phytosterols, red yeast rice with monacolin K (i.e., natural lovastatin), and berberine (13,14). While these agents may have a role in individuals at lower global cardiovascular risk, for high-risk and very-high-risk patients, nutraceuticals should be regarded, at best, as adjunctive to pharmacotherapy. There may also be issues regarding safety and purity given that their availability is less well-regulated than licensed treatments (14,15). Cost merits consideration, as these over-the-counter nutraceuticals are often substantially higher in cost than generic treatments such as statins and ezetimibe.
Combination therapy for elevated triglycerides
The other area addressed by this guidance was management of elevated triglycerides (TG) in high- and very-high-risk patients.
Accumulating evidence from mechanistic, epidemiologic and genetic studies has led to a reappraisal of the role of TG-rich lipoproteins – for which TG are a biomarker – in the causal pathway for the development of ASCVD (1). Whether lowering TG with additional treatment on top of a statin reduces cardiovascular events in high-risk and very-high-risk patients, however, remains controversial. Analyses of the ACCORD Lipid Study, together with findings from other fibrate studies, suggest that there is clinical benefit in very-high-risk patients with elevated TG (usually with low plasma concentration of high-density lipoprotein cholesterol) (16).
The data for omega-3 fatty acids are also conflicting. REDUCE-IT demonstrated a significant 25% reduction in cardiovascular events in very high-risk patients (with ASCVD and/or type 2 diabetes) with high-dose icosapent ethyl, a purified eicosapentaenoic acid (EPA) ethyl ester (17). At the time of publication of the 2019 ESC/EAS guidelines, STRENGTH had not yet reported. The lack of clinical benefit with a high-dose carboxylic acid formulation of EPA and docosahexaenoic acid (DHA) observed in this trial (18) created much uncertainty about the role of omega-3 fatty acids in high and very high-risk patients. This EAS Task Force discusses the findings of REDUCE-IT and STRENGTH in context, to understand reasons for this paradox. These may relate to the choice of comparator in each trial, possibly differing biological effects of EPA and DHA, as well differences in the patient populations (19). Thus, caution is necessary before endorsing high-dose omega-3 fatty acid preparations for preventing cardiovascular events.
Before considering which TG-lowering therapy to add, there are several pertinent considerations. First, lifestyle intervention is the first fundamental step in TG management, and therefore every effort should be made to optimise adherence with dietary and physical activity recommendations (1). Second, this practical guidance relates to the management of mild to moderate hypertriglyceridaemia (<5.6 mmol/L or <500 mg/dL). Individuals with very high TG levels (>10 mmol/L or >800 mg/dL), are likely to have a multifactorial or polygenic predisposition to hypertriglyceridaemia and are at high risk of pancreatitis.
These patients should be managed with a low-fat diet (< 10% of calories from fat) with high-dose omega-3 fatty acids, although fibrates may prove useful in polygenic chylomicronaemia (20).
Beyond lifestyle intervention, statin treatment is the first option for managing elevated TG. If this is inadequate, combination with either a fibrate or omega-3 fatty acids (high-dose icosapent ethyl, based on REDUCE-IT) may be considered. Additionally, for high-risk and very-high-risk patients with type 2 diabetes, effects beyond macrovascular benefit may be relevant to treatment decisions. Thus, the addition of fenofibrate may be considered in these patients (with or without ASCVD) with TG >200 mg/dL or 2.3 mmol/L on statin treatment, given evidence of microvascular benefits in ACCORD Lipid and other trials (Figure 3) (21).
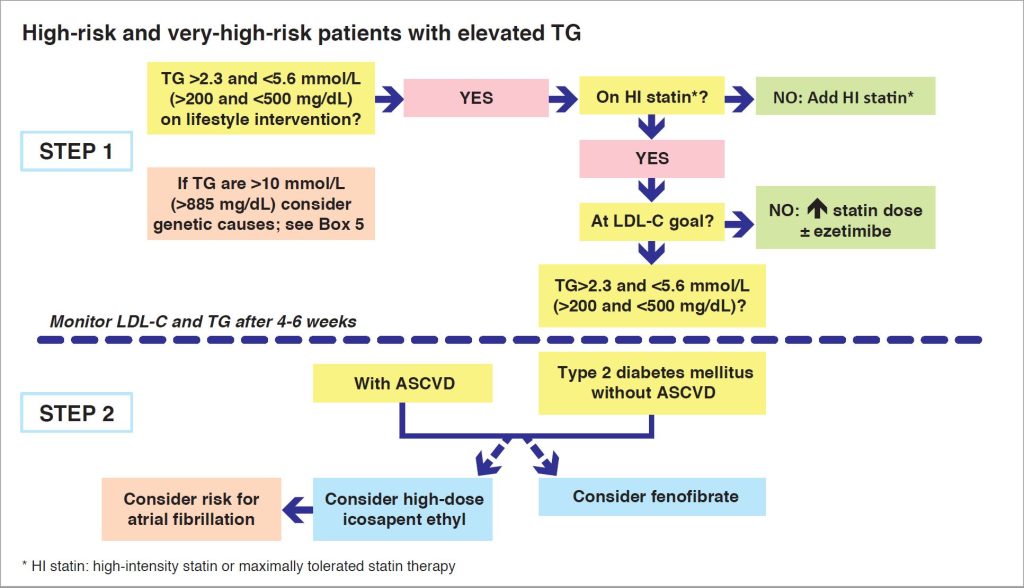
Figure 3. Algorithm for managing elevated TG in high and very-high risk patients
Conclusion
This timely statement from this EAS Task Force provides much-needed practical guidance for the use of combination therapy to manage elevated LDL-C and/or TG in high-risk and very-high-risk patients. The statement uses an accessible approach to address key areas of uncertainty.
As for the management of other major cardiovascular risk factors, a sea change in thinking is needed for the management of dyslipidaemia. Clinicians should consider combination lipid-modifying therapy as routine in high-risk and very-high-risk patients to improve LDL-C goal attainment. Implementation of the practical guidance provided by this EAS Task Force statement should be a priority, given the tantalising promise of novel agents for managing both dyslipidaemias in the future.
References
1. Mach F, Baigent C, Catapano AL, et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J 2020;41:111-88.
2. Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med 2015;372:2387-97.
3. Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med 2017;376:1713-22.
4. Schwartz GG, Steg PG, Szarek M, et al. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med 2018;379:2097-107.
5. Giugliano RP, Pedersen TR, Park JG, et al. Clinical efficacy and safety of achieving very low LDL-cholesterol concentrations with the PCSK9 inhibitor evolocumab: a prespecified secondary analysis of the FOURIER trial. Lancet 2017;390:1962-71.
6. Ray KK, Molemans B, Schoonen WM, et al. EU-Wide Cross-Sectional Observational Study of Lipid-Modifying Therapy Use in Secondary and Primary Care: the DA VINCI study. Eur J Prev Cardiol 2020; doi: 10.1093/eurjpc/zwaa047.
7. Packard CJ, Chapman MJ, Sibartie M, et al. Intensive low-density lipoprotein cholesterol lowering in cardiovascular disease prevention: opportunities and challenges. Heart 2021; Epub ahead of print.
8. Annemans L, Packard CJ, Briggs A, Ray KK. ‘Highest risk-highest benefit’ strategy: a pragmatic, cost-effective approach to targeting use of PCSK9 inhibitor therapies. Eur Heart J 2018;39:2546-50.
9. Averna M, Banach M, Bruckert E, et al. Practical guidance for combination lipid-modifying therapy in high- and very-high-risk patients. A Statement from a European Atherosclerosis Society Task Force. Atherosclerosis 2021; DOI:https://doi.org/10.1016/j.atherosclerosis.2021.03.039
10. Stroes ES, Thompson PD, Corsini A, et al. Statin-associated muscle symptoms: impact on statin therapy-European Atherosclerosis Society Consensus Panel Statement on assessment, aetiology and management. Eur Heart J 2015;36:1012-22.
11. Landmesser U, Chapman MJ, Stock JK, et al. 2017 Update of ESC/EAS Task Force on practical clinical guidance for proprotein convertase subtilisin/kexin type 9 inhibition in patients with atherosclerotic cardiovascular disease or in familial hypercholesterolaemia. Eur Heart J 2018; 39:1131-43.
12. Jenkins DJ, Jones PJ, Lamarche B, et al. Effect of a dietary portfolio of cholesterol-lowering foods given at 2 levels of intensity of dietary advice on serum lipids in hyperlipidemia: a randomized controlled trial. JAMA 2011;306:831-9.
13. Banach M, Bruckert E, Descamps OS, et al. The role of red yeast rice (RYR) supplementation in plasma cholesterol control: A review and expert opinion. Atheroscler Suppl 2019;39:e1-e8.
14. Poli A, Barbagallo CM, Cicero AFG, et al. Nutraceuticals and functional foods for the control of plasma cholesterol levels. An intersociety position papers. Pharmacol Res 2018;134:51-60.
15. Fogacci F, Banach M, Mikhailidis DP, et al. Safety of red yeast rice supplementation: A systematic review and meta-analysis of randomized controlled trials. Pharmacol Res 2019;143:1-16.
16. Ginsberg HN. The ACCORD (Action to Control Cardiovascular Risk in Diabetes) Lipid trial: what we learn from subgroup analyses. Diabetes Care 2011;34(suppl 2): S107-S108.
17. Bhatt DL, Steg PG, Miller M, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med 2019;380:11–22.
18. Nicholls SJ, Lincoff AM, Garcia M, et al. Effect of high-dose omega-3 fatty acids vs corn oil on major adverse cardiovascular events in patients at high cardiovascular risk: The STRENGTH randomized clinical trial. JAMA 2020;24:2268-80.
19. Sharma G, Martin SS, Blumenthal RS. Effects of omega-3 fatty acids on major adverse cardiovascular events: what matters most: the drug, the dose, or the placebo? JAMA 2020;324:2262-4.
20. Hegele RA, Borén J, Ginsberg HN, et al. Rare dyslipidaemias, from phenotype to genotype to management: a European Atherosclerosis Society task force consensus statement. Lancet Diabetes Endocrinol 2020;8:50-67.
21. Chew EY, Ambrosius WT, Davis MD, ET AL. Effects of medical therapies on retinopathy progression in type 2 diabetes. N Engl J Med 2010;363:233-44.